A fascinating phenomenon involving many organic molecules, and among them many biomolecules, is their ability to spontaneously form ordered structures. This process, called self-assembly, gives rise to an amazing wealth of structures of different dimensionality, determined by the balance between energy and entropy.
Biomimetic membranes
The cell membrane is a very thin envelope, a few nanometers thick, which acts as a barrier between the cell and the external environment. The exchange of information or material between the cell and its external environment is regulated by the membrane. A large number of biomolecules contribute to the functioning of the membrane, but the membrane structure is based on a lipid bilayer. Lipid membrane models, which can be experimentally obtained by exploiting the self-assembly process, are a fundamental tool for studying processes involving the lipid matrix of the membrane.
We use fluorescence or atomic force microscopy and spectroscopy to study physiological and pathological processes not yet fully understood, such as membrane fusion, or permeabilization induced by external agents, including ions, molecules of pharmacological interest or associated with pathologies, nanoparticles. We are also able to obtain information on the structural and mechanical properties of membranes and on the changes in these properties induced by external agents.
Biosensors
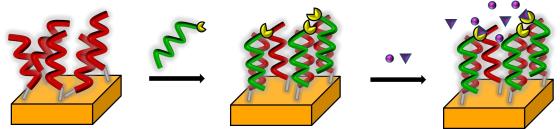
The development of biosensors is largely based on the design of hybrid systems obtained by coupling biomolecules and inorganic surfaces. Protein microarrays and sensors based on functionalized nanoparticles are just two examples among the many recent developments in nanobiotechnology.
The functionalization of inorganic surfaces, such as metals or oxides, with biomolecules is a suitable strategy to develop hybrid interfaces that present the typical functional properties of the deposited biomolecules. The self-assembly of biomolecules on surfaces is a complex process that depends on the interplay between molecule/molecule, molecule/surface, and molecule/solvent interactions. The aim of our activity is to understand the mechanisms of interaction between biological molecules and surfaces in order to design selective platforms that, exploiting the molecular recognition properties of biomolecules such as DNA or proteins, are able to selectively detect biomarkers, overexpressed in specific diseases.
Biomaterials
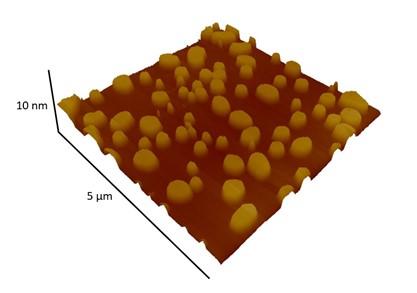
A further research activity that falls within the biomolecule/surface topic concerns the study of micro/nano-structured metal oxide surfaces of interest in the biomedical field for the development of materials for prosthetic implants. Two important properties for prosthetic materials are the resistance to bacterial adhesion and the ability to promote osteointegration. This activity is therefore focused on the preparation of porous transition metal oxide films and their functionalization with proper organic/bio-molecules that promote antibacterial properties and osteointegration.



